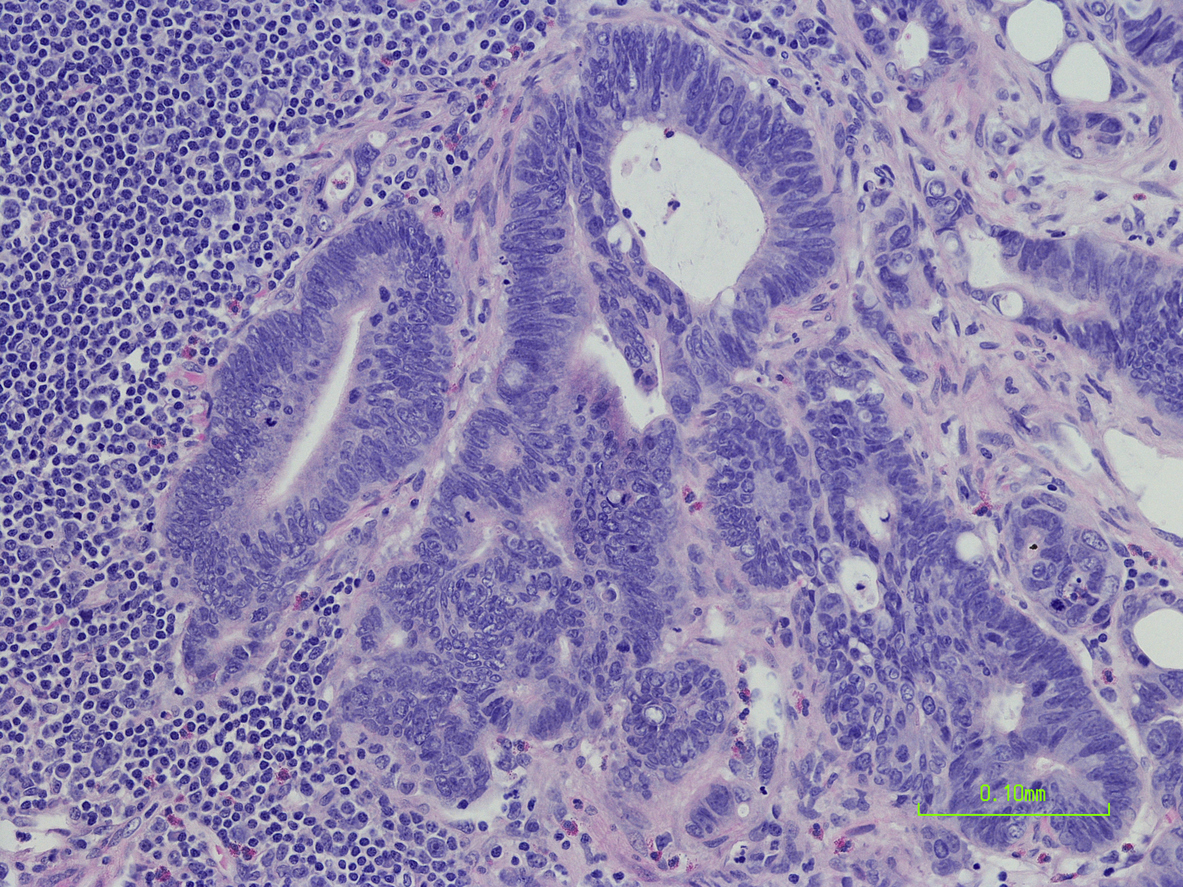
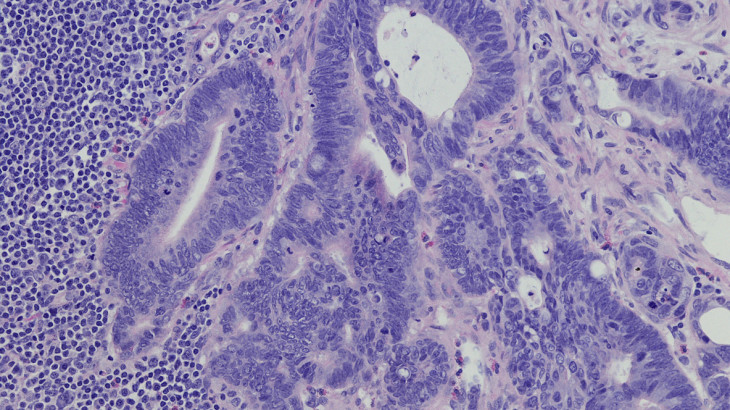
Program Overview
This continuing education activity will focus on recent clinical trial data and scientific advances in the treatment and management of patients with metastatic colorectal cancer as presented at the 2018 Gastrointestinal Cancers Symposium held in San Francisco, California. Three expert faculty will review the science of colorectal cancer, including patient screening guidelines and molecular characterization. Recent clinical trial data of emerging first-line, second-line and combination therapies for the treatment of metastatic colorectal cancer will be discussed. The faculty will also review how to best utilize the interprofessional team to create optimal treatment strategies to improve outcomes and the overall quality of life in patients with colorectal cancer. Upon completion of this educational activity, clinicians will gain better insight on the novel therapeutics for the treatment of metastatic colorectal cancer.
Target Audience
Oncologists, internists, nurse practitioners, nurses, and other healthcare professionals involved in the management of patients with colorectal cancer.
Learning Objectives
1. Discuss the science of colorectal cancer, including patient screening guidelines and molecular characterization.
2. Analyze clinical trial data of emerging first-line, second-line and combination therapies for the treatment of metastatic colorectal cancer presented at ASCO GI 2018.
3. Utilize the interprofessional team to create optimal treatment strategies to improve outcomes and the overall quality of life in patients with colorectal cancer.
Faculty
 Jeffrey A. Meyerhardt, MD, MPH
Jeffrey A. Meyerhardt, MD, MPH
Douglas Gray Woodruff Chair in Colorectal Cancer Research
Clinical Director, Gastrointestinal Cancer Center
Deputy Clinical Research Officer
Dana-Farber Cancer Institute
Associate Professor of Medicine
Harvard School of Medicine
Boston, MA
Michael Casey, APRN-BC, FNP
Dana-Farber Cancer Institute
Harvard Medical School
Boston, MA
Andrea Cercek, MD
Assistant Attending Physician
Gastrointestinal Oncology Service
Division of Solid Tumor Oncology
Department of Medicine
Memorial Sloan Kettering Cancer Center
New York, NY
Disclosures of Conflict of Interest
It is the policy of AcademicCME that all faculty, instructors, and planners disclose real or apparent conflicts of interest relating to the topics of this educational activity.
The faculty reported the following financial relationships or relationships to products or devices they or their spouse/life partner have with commercial interests related to the content of this CE activity:
| Faculty | Relationship Identified With: |
| Jeffrey A. Meyerhardt, MD, MPH | Consultant/Advisor: Ignyta |
| Michael Casey, APRN-BC, FNP | Nothing to disclose. |
| Andrea Cercek, MD | Nothing to disclose. |
Planners, Managers, Reviewers
Timothy Hayes, MD, PhD; Emma Boring; Chelsey Benedek and Kim Cheramie, MSN, RN-BC hereby state that they or their spouse/life partner do not have any financial relationships to products or devices with any commercial interests related to the content of this activity of any amount during the past 12 months.
Accreditation Statement
AcademicCME is accredited by the Accreditation Council for Continuing Medical Education (ACCME) to provide continuing medical education for physicians.
AcademicCME is accredited as a provider of continuing nursing education by the American Nurses Credentialing Center’s Commission on Accreditation
Credit Designation Statement
AcademicCME designates this enduring material for a maximum of 1.0 AMA PRA Category 1 Credit(s)™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
AcademicCME designates this enduring material for a maximum of 1.0 CNE contact hour.
Financial Support
This activity has been supported by an independent educational grant from Amgen Inc.
Disclosure of Unlabeled Use
This educational activity may contain discussion of published and/or investigational uses of agents that are not indicated by the FDA. AcademicCME and Amgen, Inc. do not recommend the use of any agent outside of the labeled indications.
Disclaimer
Participants have an implied responsibility to use the newly acquired information to enhance patient outcomes and their own professional development. The information presented in this activity is not meant to serve as a guideline for patient management. Any procedures, medications, or other courses of diagnosis or treatment discussed or suggested in this activity should not be used by clinicians without evaluation of their patient’s conditions and possible contraindications on dangers in use, review of any applicable manufacturer’s product information, and comparison with recommendations of other authorities.
Method of Participation
- Read the learning objectives, accreditation information and faculty disclosures at the beginning of this activity.
- Complete the Pre-Activity Questions.
- Read or Review the activity content.
- Complete the Post-Activity Test Questions and Evaluation.
- Healthcare providers who receive a grade of 70% or better on the Post-Activity Test Questions and who complete the Evaluation will receive a CME/CNE Certificate.
- All other participants who receive a grade of 70% or better on the Post-Activity Test Questions and who complete the Evaluation will receive a Certificate of Participation.
- Healthcare providers should claim only the credit commensurate with the extent of their participation in the activity.
- 60 minutes of participation = 1 CNE contact hour
CE Inquiries/Special Needs
For all CME/CNE inquiries or special needs, please contact [email protected].